%22NAC%22 Supplementation and COVID-19 (N-Acetylcysteine) (Lecture 69)
%22NAC%22补充剂和COVID-19; N-乙酰半胱氨酸 (新冠讲座69) – 本文由 ‘中国推动’ 学者、吉林大学医学院毛宗韬同学编辑整理
Welcome to another medcram covid-19 update where over four million total confirmed worldwide at the very least. Let’s take a look at some of these countries. And while the United States tops the list in terms of total cases. If you look at Deaths/1M pop, we’re certainly not near the top of that. Europe is still at the top of that list.
In terms of Spain, the number of new cases per day is trailing off and that’s holding true for the number of new deaths per day in Spain as well. The number of cases in Russia on a daily basis have also increased here recently, and that in addition to the number of deaths as well.
And when we look at a country like India, very large, we’re also seeing here that the number of cases is going up, doesn’t look like they have peaked yet. And the number of daily deaths is also going along with that as well at this point. And we’re all keeping our eyes on Australia. I know they’re supposed to be headed into winter there at this point, but it’s still pretty warm. So, we’ll have to see how things go once the weather starts to cool down there in Australia. And they haven’t had deaths there in Australia for some time now. We haven’t heard from South Korea in some time and they’ve been doing a tremendous job at keeping the number of new cases at bay here.
Well, today we’re going to go back to medical school a little bit and we’re going to talk more about our oxidative stress. Because I think this is really important and we’ve been building on this for the last week or so. In terms of the lectures, once again, we have oxygen, and when you take an oxygen molecule and add an electron to it, and you get a superoxide. You add another electron to it, you get two oxygen molecules and two hydrogen molecules attached to it. Add another electron molecule, you got a hydroxyl and then finally you get water. And so, the most reduced form here is the H2O or the water, and the most oxidized form here is O2. Also, please realize that when you add electrons, protons come on as well. And that’s why you start to see the ratio of protons to oxygen molecules increase as you go from left to right. The body’s defense mechanisms are again superoxide dismutase (SOD) which converts the superoxide form here into hydrogen peroxide (H2O2) and oxygen. There’s also catalase which converts hydrogen peroxide into oxygen and into water. And then, finally the one that we’re going to talk a little bit more about is this glutathione peroxidase (GSHPX) and I want you to notice here that glutathione peroxidase takes glutathione (GSH), which is reduced. And it reduces the hydrogen peroxide and turns it into water while itself becomes oxidized into this disulfide bond is what it’s called. And so, the sulfide attached to the hydrogen is the reduced form. And the sulfite attached to another sulfide is the oxidized form that in turn gets regenerated back by taking the reduced form of NADP+, which is NADPH, and converting it into NADP+ through this enzyme called glutathione reductase. So, there’re other ways of reducing glutathione from its disulfide bond form into its sulfhydryl group.
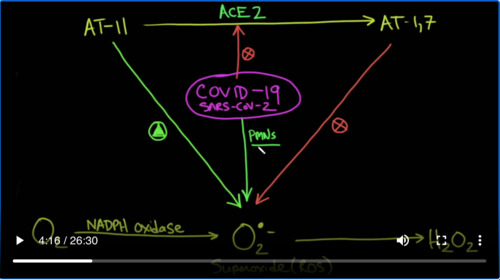
And again, the reason why this is important is because we’ve shown how superoxide can accumulate in oxidative stress environments such as diabetes, hypertension, coronary disease and that when you have covid-19 that sets up a perfect storm of superoxide. Why is that? Once again, it’s because covid-19 hits the ACE2 enzyme which is its receptor. And therefore, there’s an accumulation of AngiotensinⅡ (ATⅡ) which stimulates the production of superoxide. There’s also a deficiency of Angiotensin1-7 (AT1-7) which inhibits the production of superoxide through a nitric oxide mediator. In addition to this covid-19 itself attracts PMNs which are neutrophils which also can cause production of superoxide.
And that’s super oxide which is right here is going to cause production of hydrogen peroxide and a whole bunch of other hydroxy radicals, which can cause oxidation and destruction of endothelial cells and possibly even thrombosis.
So this is glutathione. And for those of you who are biochemically inclined, you’ll see the backbone here of an amino acid chain N-C-C-N-C-C. And you’ll notice that this, right here is the amino acid cysteine and that’s where N-acetyl cysteine (NAC) would come from but it also can be regenerated glutathione by N-acetylcysteine, which we’ll talk about in a little bit. The key point that I want you to see however is this SH. This is really important because this SH can also bond with another form of this molecule and instead of having an S bound to an H. It can bind to another S that would be the oxidized form. Here this is in the reduced form. There’s a hydrogen bonded to this sulfur group. This sulfur group can bind to another sulfur group and that would be the oxidized form. So, what does that mean? Well, what it means is that if you’ve got 2 glutathione with this SH group and you oxidize it with hydrogen peroxide, you will couple two of this glutathione together in a bond that will make them connected and you’ll release water. So, notice, 2 reduced glutathione plus hydrogen peroxide gives you an oxidized glutathione plus water.
And that should look familiar. Again, here, we have 2 glutathione being oxidized to a connected disulfide bond in which you get is hydrogen peroxide turning into water.
That’s important to understand, because here you have a G-S-H.
and what you’re doing is you’re going to a G-S-S-G.
2 independent molecules now get connected.
And I’m going to plant a little seed in your head with this article, which says that disulfide bond reduction of von Willebrand factor (VWF) by ADAMTS-13. That’s right. Von Willebrand factor actually comes together and polymerizes with helped in part with disulfide bonds. By the way, this is the same thing that happens with hair. So, hair is simply strands of keratin, which is the spiral structure, and a single thread of hair is basically multiple keratin fibers. And what’s happened is their S groups that are binding these together. And then what happens when you go for a perm is it’s basically a reducing agent and you break all of these disulfide bonds and you put Hs on them and then you bend the hair so it’s like this. And then what happens is you neutralize the perm agent by putting hydrogen peroxide on the perm agent. And what happens is the new disulfide bonds will form keeping the hair with its curl better living through chemistry.
So, if in fact covid-19 gives us an excess of superoxide and hydroxy radicals, and that excess of superoxide can in fact be taken care of through things like glutathione peroxidase. Are the things that we can do to help glutathione peroxidase be in the reduced form to take care of this, so we can alleviate the stress from this oxidative stress? And the answer is potentially yes.
Enter N-acetylcysteine (NAC), otherwise known as NAC or “N-A-C”. As you can see, N-acetylcysteine is both an antioxidant and a disulphide breaking agent, and here’s the reason why. You can see very clearly the fact that NAC has a sulfur bound to a hydrogen meaning it’s a reducing agent. And it looks very similar to cysteine (Cys) its amino acid except it has an acetyl group bound to the nitrogen. That’s why it’s known as N-acetylcysteine. And as you can see here very clearly, NAC will break up these disulfide bonds and put hydrogens on them in the reduced state so that these strands will no longer be connected. So, these highly cross-linked and elastic mucus gels get broken up and you get reduced viscosity. And that’s exactly how NAC used to be used inhaled for people who wanted to cough up sputum that was thick. It was a mucolytic. The other thing that NAC would do is it would take the metabolite of Tylenol. If Tylenol overdose was a concern, this metabolite from the Tylenol would be here and it would over utilize and use up all your glutathione. Well, what N-acetylcysteine would do is you would take off the acetyl group and you’d be left with cysteine. And cysteine would recharge glutathione and allow you to metabolize the toxic agent and prevent the cell damage and the immune response that is all too well known in Tylenol overdose from liver toxicity. As you can see here, NAC acts by replenishing hepatic glutathione, which is the main endogenous nucleophilic peptide able to neutralize the major metabolite of acetaminophen, which is Tylenol. Now in Europe is known as paracetamol and it’s metabolized forming the electrophilic metabolites and NAPQI which is detoxified by glutathione. In the case of glutathione depletion, and NAPQI reacts with proteins here forming adducts which can induce cell damage or immune response. And that’s how you get liver failure and death. So, you can see that NAC is a very important molecule, not only is it favorable for saving people from Tylenol overdose, but it can also act as a mucolytic as you can see here. But that’s not all that NAC is useful for. The article goes on to talk about how glutathione is used in many different reactions in the cell. And it can become depleted especially in some regular type of infections like colds and tonsillitis Etc.
It says here that they performed a review of the literature from 1980 to 2016 on the role of oxidative stress and glutathione play in the ear, nose, and throat (ENT) conditions. The authors concluded that many ENT conditions such as rhinitis, allergic rhinitis, chronic rhinosinusitis (CRS), CRS with polyps, otitis media with effusion, chronic otitis media (COM), COM and cholesteatoma, tympanic membrane sclerosis, tonsillitis, Meniere’s disease, laryngeal conditions, and chronic cough are associated with you guessed that oxidative stress and decrease glutathione. Both locally in the affected tissues and systemically decrease glutathione levels, are also associated with common features of aging as well as a wide range of pathologic conditions. So, it seems safe to say here that glutathione could be recharged with N-acetylcysteine.
Well, let’s see if it actually works in diseases. Now, here’s a study that was published in 1997, looking at a total of 262 subjects with both sexes. This was a randomized double-blind trial looking at over 20 different centers in Italy. And they were randomized to look at either Placebo or NAC tablets (600 mg) twice daily for six months. And when they looked at how many people came down with the H1N1 flu, there was no difference. Both groups came down with the same amount of flu. But when they looked at which one actually had symptomatic forms of the flu, the placebo group 79% of them came down with symptomatic forms of the flu, which is only 25% of those with NAC treatment. Wow, that’s an absolute risk reduction of 5%, which means you’d only need to treat two people to prevent one of them from getting the flu symptoms. So they concluded that administration of N-acetylcysteine during the winter appears to provide a significant attenuation of influenza and influenza-like episodes, especially in elderly high–risk individuals. Now, it didn’t prevent the influenza infection, but it’s significantly reduced the incidence of clinically apparent disease.
Here’s another article that was published in 2010 showing that the same substance NAC, actually inhabited virus replication and reduce these inflammatory cytokines and these pro-inflammatory molecules. And the recommendation was that therefore antioxidants like NAC represent a potential additional treatment option that could be considered in the case of an influenza A virus pandemic.
Now here’s a meta-analysis looking at the effects of N-acetylcysteine treatment in acute respiratory distress syndrome of all types. This was a meta-analysis again in 2017. Now remember that the definition of ARDS had changed and so the number of trials here maybe a bit heterogeneous. But what they found was that there was a statistically significant difference in the length of ICU stay even though there wasn’t a difference in the 30-day mortality or the short-term mortality. And they left by saying as the number of included trials in patients with small additional trials are required to provide sufficient evidence for the efficacy of NAC in ARDS. But as part of their criteria and as their rationale, they do say here that removal of reactive oxygen species by antioxidants has been applied in clinical practice and that NAC is the most commonly used antioxidants. Another key finding is that no severe adverse reactions were observed in the patients that were included.
Here’s another article. This one published in 2018 titled N-acetylcysteine Improves Oxidative Stress and Inflammatory Response in Patients with Community-acquired Pneumonia. And what they did here was they looked for different levels of oxidative stress. And what they found were these levels of oxidative stress were similar between the two groups before treatment, but that plasma levels of MDA and tumor necrosis factor-α (TNF-α) decreased more in the NAC group than in the non-NAC group, including total antioxidant capacity (TAOC). Again here, they said that no NAC-related adverse effects were observed. Of course, this is for community-acquired pneumonia, not covid-19. It may be that oxidative stress plays more of a role in covid-19 than it does in community-acquired pneumonia. In fact, it might play a great role. Because as we’ve shown before, this oxidative stress may lead to thrombosis from hypercoagulable state and from von Willebrand’s factor. There may be another front that N-acetylcysteine is opening up here with covid-19 in that. And acetyl system may not only affect the oxidative stress aspect of covid-19, but also the hypercoagulable state that is set up by the excess of von Willebrand faster.
Here, we have this article that was published in 2013, which shows that N-acetylcysteine can have an effect on the coagulation factors in plasma samples from healthy subjects. They concluded that they were able to demonstrate a significant decrease in the activity of coagulation factors Ⅱ, Ⅶ, Ⅸ and Ⅹ with the addition of N-acetylcysteine. This would mean that the patient’s blood would be thinner or less apt to clot.
This paper was published in 2006 looking at the effect of N-acetylcysteine on blood coagulation and platelet function in patients undergoing open repair of abdominal aortic aneurysm. And what they concluded was that NAC has anticoagulant and platelet–inhibiting properties in patients undergoing major vascular surgery. This abnormal hemostatic activity should be considered when NAC is administered to patients with increase bleeding risk.
And the articles just keep coming. This one published two years ago. They said they previously demonstrated that diabetes exacerbates stroke–induced brain injury, and that this correlates with brain methylglyoxal (MG) -to-glutathione (GSH) status. Again, our friend glutathione. Cerebral injury was reversed by N-acetylcysteine (NAC). Here we tested if the prothrombotic phenotype seen in the systemic circulation and brain during diabetes was associated with increased MG-glycation of proteins and if NAC could reverse this. So, what they did was they gave NAC. And this is what they showed. They said collectively, these results show that the diabetic blood and brain become progressively more susceptible to platelet activation and thrombosis. I would have to assume that this might be through the same process of oxidative stress. NAC, given after the establishment of diabetes, may offer protection against the risk of Stroke by altering both systemic and vascular prothrombotic responses via enhancing platelet GSH, and GSH-dependent MG elimination, as well as correcting levels of antioxidants such as superoxide dismutase one (SOD-1) and glutathione peroxidase one (GPX-1). These are both enzymes that are specifically discussed here on our videos.
And now the best paper yet. This one published just three years ago. Now remember what we said about oxidative stress in covid-19. We thought that the oxidative stress was causing endothelial damage. And that endothelial damage was causing the release of von Willebrand factors, which was cross-linking and allowing thrombosis to occur and the hypoxemia, perhaps in covid-19. Well, here’s a paper titled Potent Thrombolytic Effect of N-acetylcysteine on Arterial Thrombi. Let me just say Circulation is a very prestigious journal. Certainly, it is peer reviewed. And what they say here is that platelet cross-linking during arterial thrombosis involves von Willebrand factor multimers. Therefore, proteolysis that means breaking up of von Willebrand factor appears promising to disaggregate platelet-rich thrombi and restore vessel patency in acute thrombotic disorders such as ischemic stroke, acute coronary syndrome, or acute limb ischemia. NAC or N-acetylcysteine can reduce intrachain disulfide bonds. There it is. Remember what we said that N-acetylcysteine can actually break up these S–S bonds. Well, here is a place where it might be able to help in terms of breaking up these clots in large polymeric proteins. In the present study, we postulated that NAC might cleave the von Willebrand factor multimers inside of occlusive thrombi, thereby leading to their dissolution and arterial recanalization. That means opening up the arteries once again. So, what did they find? Exactly that NAC administration promotes lysis of arterial thrombi. These are the same kind of thrombi that might be acting in covid-19 in the pulmonary vasculature and in other places as well. They found that the thrombolytic effects of NAC is principally at the von Willebrand factor. And that if you administer it with a GPⅡb/Ⅲa inhibitor like Integrilin which is another medication that it actually improved its thrombolytic effect even more. That this was not accompanied by significant worsening hemorrhagic stroke outcomes, suggesting that it exerts thrombolytic effects without significantly impairing normal hemostasis. We provide evidence that NAC is an effective and safe alternative to currently available antithrombotic agents to restore vessel patency. Fascinating!
And here is another article that was actually just published earlier this year, again showing the same thing that N-acetylcysteine inhibits thrombosis in a murine model of myeloproliferative neoplasm.
Here is another article published in 2011 showing that NAC reduces the size and activity of, you guessed it, von Willebrand factor in human plasma and in mice. And here you can see in the journal of Clinical Investigation, these long multimers of von Willebrand factor. And you can see in the control, the number of strings versus that in NAC. So basically, it cuts these things up. So, you may ask, well, how come no one’s talked about and ask the tail system before.
Well, here’s a review that was done last month on April 14th titled N-acetylcysteine: A Rapid Review of the Evidence for Effectiveness in treating Covid-19. And they do go through a very nice review where they look at NAC for acute respiratory disorders. But again, this is before we really understood that maybe covid-19 is not just a respiratory disorder, but it’s maybe also a vascular disorder causing thrombosis, which is causing a lot of the hypoxemia. So they did look at NAC in terms of acute respiratory disorders. And the verdict that they came up was that clinical trial evidence for the use of NAC as an antioxidant in influenza and other acute viral respiratory tract infections is very limited and therefore difficult to draw any concrete conclusions without further trial evidence. And so, they’d both through the data here in terms of its antioxidant properties, but they really don’t touch at all on NAC and its ability to cleave those disulfide bonds and breakup and thrombolysis some of these clots that we are seeing. And it’s well referenced.
So, when I went to ClinicalTrials.gov to see if anything was being done, I did find only one study: A study of N-acetylcysteine in Patients with Covid-19 Infection. And if you look here, they’re actually giving it to patients who have severe disease, which was very happy to see, they are trying to enroll 86 participants. It was a non–randomized trial and it was a phase Ⅱ study of N-acetylcysteine in severe or critically ill patients with refractory covid-19 infection. And they’re not fooling around folks because they’re giving 6 g/day of IV N-acetylcysteine and they’re going to give it up to 3 weeks in duration or until they’re discharged from the hospital, admitted to a critical–care unit, intubation, toxicity or death.
Let’s take a look at our hypothesis one more time. So we got SARS-Cov-2 that comes in, binds the ACE2 receptor thereby taking it out of action. So you have reduced ACE2. As a result of that, your Angiotensin Ⅱ increases and your Angiotensin 1–7 decreases that causes an increase in superoxide. That increase in superoxide causes oxidative stress, which causes endothelial cell dysfunction which increases the amount of von Willebrand factor released from the subendothelial space and that causes thrombosis. And potentially it’s the thrombosis that’s causing the hypoxemia and other things in the pulmonary system. So, the nice thing about N-acetylcysteine is that it could act here to reduce the oxidative stress and it can potentially act here to reduce the amount of von Willebrand factor monomers and polymers coming together, and forming these clots and the thrombosis that is associated with it. Again, we have no evidence that this would work in covid-19 because we haven’t done the trials. But if we connect the dots, it looks promising and more importantly in a number of studies where they gave NAC, they did not see serious adverse outcomes. So, the question that I have is, if people took NAC orally as a preventative, could this prevent serious forms of covid-19, just like we saw with influenza A, it didn’t reduce the amount of infections, but it reduces the severity of the infections? The next question is, if given in high enough doses intravenously in the hospital, can it prevent the patients from progressing to intubation and intensive care admission? Thanks for joining us.



Add comment