Update 79: Five Vaccines to Keep an Eye On
medcram.com welcome to another medcram covid-19 update
last time we talked about vaccines and the different phases that they were in and we were referred you to this vaccine tracker. What I wanted to do was narrow it down a little bit more into about five vaccines to keep your eye on in the future. It’s these five that have the most likely chance of being available in the early part of 2021. And the first one is the one by moderna. It’s the MRNA 1273.
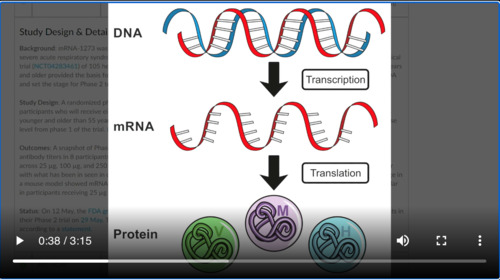
This one’s a special one in that. It’s an mRNA vaccine, which means that the MRNA is going to be put into person’s body and that’s going to generate the protein which then will generate the immune response. Now, you have to remember there are three phases to any kind of drug trial or vaccine phase one is a safety issue Phase 2 looks a little bit more at dosing and then phase 3 is the pivotal trial if you will where the actual data comes out to see whether or not it’s working. They’ve already gotten into phase two trials here.
29th of May and the phase 3 trials are expected to start in the early summer. So they’re already on pretty good time tracks. The next vaccine is the University of Oxford vaccine here. This vaccine is looking at a different type of viruses in adenovirus vaccine Vector, but it’s going to inject the epitope or the antigen into the patient. This team is actually come together and made the MERS vaccine in the past. So this is a pretty promising group and they already started
recruitment for Phase 2 3 trials here on May 22nd. And so this is another one to be looking for.
Here’s the third one Johnson & Johnson. They’re still in preclinical trials and they are estimating that Phase 1 trials are going to start in September of 2020.
The fourth one is Merc. They’re also in preclinical and they are using technology that’s allowed them to come up with the Ebola vaccine. And finally the last one is Pfizer. This is also looking at it am rna-based vaccine and this is already been in Phase 1 and two trials since April 22nd.
Of course the one that’s gotten the most media attention is the one from moderna. What’s interesting about this study. Is that the FDA gave permission to the regulatory body to allow moderna to start doing these trials without safety data from animals first.
As you can see here in this article titled researchers brush to test coronavirus vaccine in people without knowing how well it works in animals this article talks about some of the ethics involved with that and realizing that this is how were able to get these vaccines so quickly and if you want to hear more about vaccines and what we’re doing with them in covid-19, I wanted you to know about a live Symposium that we’re having today on Med IQ.
I plan on being with Dr. Desai and there will be a link in the description below. So quick video today and we’ll see a live this afternoon. Thanks for joining us.



Add comment