Update 75: COVID-19 Lung Autopsies
Welcome to another Medicram COVID-19 update. It’s Memorial Day here in the United States. I am just finishing up about eight days of intensive care service. And at the hospital that I work at. we actually have created a second complete ICU. We have our original ICU, which we continue to see patients that don’t have COVID-19 and we’ve opened up a completely different section of the hospital, which is an ICU dedicated to treating all positive COVID-19 patients and both are full. We’ve essentially doubled the amount of ICU patients we have in the hospital. This is not counting COVID-19 patients that are on the regular floor not in the Intensive Care Unit and what I’m finding out in terms of taking care of these patients is what we might get maybe one or two patients a day into this kind of a unit, maybe one or two patients every couple of days into this unit. These are patients that have come in there on the floor. They’re being treated with numerous medications. All combinations of different medications that we’ve talked about here depending on what the physician feels is appropriate for the patient and for whatever reason some get better, but a number of them get worse. They continue to get worse and finally they require to be placed on a ventilator even though we try to hold off on putting these patients on ventilators as long as we possibly can. We’ll put these people on ventilators not because their oxygen is low, but because they’re breathing so hard and so fast that they simply just can’t keep up anymore. And they’re wearing themselves out and they just need the support of a ventilator. Once they become intubated and they’re on the ventilator. They stay on the ventilator for a very long time and they recover very very slowly. And so we accumulate these patients in our COVID-19 Intensive Care Unit. And I would say over the last month or so we’ve grown from about three patients in the Intensive Care COVID-19 Unit to now up to eight or nine patients on the ventilator in the Intensive Care Unit.
So it’s going to be interesting where this goes because the hospital that I work at is in San Bernardino County and I’m sure this is happening in many other counties across the United States and even the world. And our county it was approved just recently to move into an accelerated phase 2, open dine-in restaurants and mall phase and so as it says here “Gyms, hair and nail salons, barber shops, movie theaters, sports and entertainment venues, libraries, bars, and wineries, hotels and motels and public swimming facilities will still remain closed but there are certain entities that will open up and serve.
And this was stated by the Board of Supervisors of San Bernardino County chairman Kurt Hagman said San Bernardino County businesses and residents worked very hard. It made tremendous sacrifices to make this moment possible. And basically how this works is that the state of California had a criteria that they had to meet before they could go into phase 2 opening and they submitted a revised request and they met that phase request and so they’re going to be moving into phase 2.
So what does that mean? It means in San Bernardino County which is the largest geographical county in the United States. It says as part of this accelerated phase 2 of the state recovery plan, destination retail stores, including shopping malls, swap meets and dine-in restaurants can now reopen in San Bernardino County. Businesses that plan to reopen are required to follow state guidance detailed at and it gives the link. They also go on to say that the virus is still very present throughout our county, state and nation. We must remain vigilant by physical distancing, wearing face coverings and washing our hands often. But our goal of minimizing illness and building the capacity to protect the vulnerable, serve the sick and track the virus in our communities has been achieved. And of course those other places that we said like gyms and hair salons will be part of stages 3 and 4. They’re going to be giving additional guidance for church services and things of that nature on Monday, May 25th.
This is going to be very interesting because with the recent opening of phase 1 in the state of California, we’ve noticed a bit of an acceleration in terms of the number of COVID-19 patients we have in the Intensive Care Unit and we are getting close to capacity in terms of that second ICU don’t know if opening an additional floor is going to be possible. Everybody at our Hospital from the administration all the way down to the front staff doctors like myself.
All of the hard-working nurses that I work with today, in fact and the hard-working respiratory therapist that I did a number of Bronx with today. They’re all putting in a number of shifts that are above and beyond what’s been required and we are trying to piece together this business that we call ICU. I wanted to revisit what we’ve been talking about last couple of weeks, which is the hypothesis that we’ve been talking about. And for those of you that haven’t been following us go back and look the last couple of weeks and it’s this hypothesis of SARS-CoV-2 going to thrombosis and just to review that for you. We know that SARS-CoV-2 hits the ACE2 receptor that down regulates ACE2. Well when that happens you get an increase in AT-II and a decrease in AT-1,7 which increases oxidative stress represented here by superoxide, that superoxide causes endothelial cell dysfunction. That is the covering of the vascular system that causes an increase in inflammation.
Action releasing von Willebrand’s factor, which leads to thrombosis we’ve been talking about this for weeks.
Enter article from The Washington Post published just a few days ago, more evidence emerges on why COVID-19 is so much worse than the flu. Let’s take a look at this article because I think it’s really interesting and summarizes what we’ve been talking about. So the article goes on to say that researchers who examine the lungs of patients killed by COVID-19 found evidence that it attacks the lining of blood vessels there a critical difference from the lungs of people who died of the flu, according to a report published Thursday in the New England Journal of Medicine. This validates our hypothesis that we made weeks ago that SARS-CoV-2 is an endothelial disease.
At least in part again SARS-CoV-2 goes through this cascade which leads to thrombosis. Let’s take a look at that New England Journal of Medicine report. And of course will give you links in the description below. This is the article from the New England Journal of Medicine that was published on the same day May 21st, “Pulmonary vascular endothelialitis, thrombosis and angiogenesis in COVID-19. The New England Journal of Medicine is a prestigious journal, probably one of the most prestigious journals in the United States and what they found after examining 7 lungs obtained during autopsy from patients from COVID-19 comparing them to 7 control lungs from patients who died of ARDS secondary to influenza infection and 10–aged match uninfected control lungs. The lungs were studied with the use of seven color immunohistochemical analysis, micro–computed tomographic imaging, scan the electron microscopy, corrosion casting, etc. So what did they find? Check this out in patients who die from Covid-19-associated or influenza-associated respiratory failure, the histologic pattern in the peripheral lung was diffuse alveolar damage with perivascular T-cell infiltration. That’s no surprise. That’s what we knew already before COVID-19 came to the United States. However, the lungs from patients with COVID-19 also showed distinct vascular feature, consisting of severe endothelial injury associated with the presence of intracellular virus and disrupted cell membranes. What they’re telling you here is that this was not seen in the patients with influenza-associated respiratory failure. This practically validates the hypothesis when they did histological analysis of pulmonary vessels in patients with COVID-19. It showed widespread thrombosis with microangiopathy. Let me just unpack that for you. Blood clots with pathology in the small blood vessels. Alveolar does the air sacs in the lung capillary, that’s where they get the gas exchange. Microthrombi, these are so small. You would never be able to see these on a CAT-scan, you might be able to see them on something called a VQ scan which looks at the ratio of ventilation and perfusion.
But notice that in COVID-19 patients, they were 9 times more prevalent than they were in the patients with influenza. And this has a very very small p-value which means that the chances of this happening just by chance is extremely low goes on to say that in the lungs from patients with COVID-19 the amount of new vessel growth predominantly through a mechanism of intussusceptive of angiogenesis was 2.7 times as high as that in the lungs with patients from influenza. In other words, the lungs here are trying to bypass the congestion. This is like if you had an accident on the freeway and the lanes were congested. You and everybody else would want to get off the freeway use the surface streets and get back on the freeway. And that’s exactly what’s going on here through the creation of new blood vessels to bypass this blockage. So their conclusion showed that in our small series, vascular angiogenesis distinguish the pulmonary pathobiology of COVID-19 from that of equally severe influenza virus infection. Here’s the word that’s important “ the universality and the clinical implications of our observations require further research to define”.
Now further in the article, you can see pictures of patients lungs who have died of COVID-19 and you can see here. This is not the nice pink tissue that you would expect to find. In fact here. If you look under the microscope, you will see that these black dots all through here are all lymphocytes that have infiltrated into the lung and have made it stiffer and have caused destruction of the tissue inflammation of the tissue as a result of this immune infiltration of these lymphocytes. And here folks is the smoking gun that may exist. Let me blow this up so you can see it better.
And I want you to look here very carefully because you see these wide open air spaces. This is where air comes in that you breathe and oxygen is supposed to diffuse into the blood vessels and normally what happens. Let’s take for instance, a blood vessel here where oxygen in the air is going to diffuse past this very thin capillary membrane and go into the blood to be picked up. Now what’s happened here though is as you can see here is a capillary space that is completely filled with blood. Here is another one, this is end on though. It’s not going to be round. It’s going to be elongated because this capillary happens to be going in the plane of the slice. Here’s another one though. And what’s happened here is the space has been completely occluded with a blood clot, and it prevents the blood from flowing. This here essentially prevents any kind of oxygen here in this alveolus from diffusing into this pulmonary capillary and therefore basically causes no picking up of oxygen. Notice also that these capillary membranes are still nice and thin. In fact, these walls while they do look a little juicy to me in terms of the thickness. They’re relatively fine. So what could happen here very easily is you would have what we call our happy hypoxic. They’re sitting there their lungs are not stiff. They’re able to breathe. It’s just that their oxygen saturation is very very low. Here’s a nice capillary here. That should be empty but it’s filled with blood clot. And therefore there is congestion the blood cannot pass through here, oxygen which is in the alveolus cannot diffuse into this blood vessel and the blood vessel. Therefore does not transmit the oxygen into its lumen and therefore the red blood cell filled with hemoglobin will not pick up the oxygen and transmit it to the rest of the body.
Here in this figure they actually took a scanning electron microscope and look deep down at the endothelium in the lungs and they found actual viral particles in the extra cellular space, confirming that the virus has had gotten to the endothelial space.
They say in the article. We found greater numbers of ACE2–positive endothelial cells and significant changes in endothelial morphology, a finding consistent with the central role of endothelial cells in the vascular phase of COVID-19. And ethelia cells in the specimens from patients with COVID-19 showed disruption of intercellular junctions, cell swelling, and a loss of contact with the basal membrane. That’s important because guess what’s in between the membrane and the endothelium. Things like Von Willebrand’s factors. This sub endothelial release of Von Willebrand factor is part of our hypothesis. The presence of SARS-CoV-2 virus within the endothelial cells, a finding consistent with other studies suggest that direct viral effects as well as perivascular inflammation, may contribute to endothelial injury.
So once again, our hypothesis seems to be validated by some of the information that’s coming out, and just to be sure this is not our hypothesis but a hypothesis based on studies that we reviewed here at Medcram seems likely and so if that is the case, it would seem that you’d want to block this cascade in many different places and as we’ve discussed before N-acetyl cysteine could interrupt this superoxide part of the cascade as well as the Von Willebrand factor part of the cascade. Because Von Willebrand factor polymerizes using disulfide bonds and N-acetyl cysteine cleves those disulfide bonds.
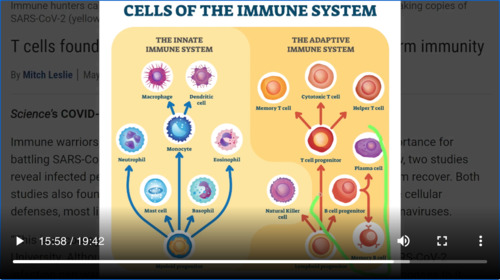
Okay, let’s move on to another article. This was an article that was published in Science and it’s an interesting article because we’ve talked about the innate immune system. But this article goes into the adaptive immune system and actually brings up some very interesting points and may have some good news for us. This is titled “T cells found in COVID-19 patients bode well for long-term immunity”. And as we’ve discussed before. The T-cell, the memory T-cell, the helper T cell, the cytotoxic T cell. These are the cells that are involved with life long immunity. And so it’s going to be important to see whether or not they response to SARS–CoV-2 involves T cells. All of the vaccines are looking at trying to make sure that B cells make antibodies and that is in fact what we’re testing is antibodies, but another important part of that adaptive immune system is the T Cell. So let’s see what they say here. They talk about immune warriors known as T cells which help up fight some viruses, but what they found two studies show that infected people with SARS-CoV-2 harbor T cells that actually target the virus—and may help them to recover. Both studies also found that some people never infected with SARS-CoV-2 also have these cellular defenses. Most likely because they were infected with other coronaviruses. Coronaviruses which share surface proteins with SARS-CoV-2.
So when they looked at people who were infected with SARS-CoV-2 or COVID-19, they found that all the patients carried helper T cells that recognized the SARS-CoV-2 spike protein. That’s the protein that the virus uses to infect cells. They also harbored helper T cells that react to other SARS-CoV-2 proteins. This what we were talking about is that there are proteins in SARS-CoV-2 that match other coronaviruses. And this goes along with another article on medical archive, which is a preprint collecting of these articles. In that study, they identified helper T cells targeting the same spike protein in 15 out of 18 patients hospitalized with COVID-19. What does this mean? It means that these patients who are getting COVID-19 are getting the response that we need to see in these long-lasting T cells that will give them life long immunity.
Now whether or not this actually translates into lifelong immunity only time will tell. But we’re seeing the things that we need to see to this ends. So the other thing I looked at is whether or not people who have never been infected with SARS-CoV-2 might have some of these features and in fact they did, because they took blood that had been stored between 2015 and 2018. Well before the pandemic began and they found that some of these patients about 34% had helper T cells that recognize SARS-CoV-2. Now, how is that even possible? Well, if you know anything about your immune system, you’ll know that there’s a T-cell out there for just about any antigen that you might come into contact with. The question is is has that T-cell been activated already by a previous infection and in fact because SARS-CoV-2 shares proteins in the coronavirus family. It’s possible that if you’ve had a cold or some other virus, some other coronavirus which shares a protein with SARS-CoV-2. It’s possible that you may have a T-cell that’s been activated against the SARS-CoV-2 virus. As it says here these researchers think that these cells were likely triggered by past infection with one of the four human coronaviruses that causes colds, proteins in this viruses resemble those of SARS-CoV-2
It may also explain why there’s a certain quote chunk of the population that’s able to deal with the infection well and why they don’t become ill
So I thought this article was really interesting because it showed us that this SARS-CoV-2 virus is acting the same way that we think it should in terms of lifelong immunity, adaptive immunity and as we’ve talked about before—innate immunity. Want to thank everybody support out there for Medcram on various social media. And once again, thank you for joining us.
Q:从生物医学角度,新冠病毒到底是如何作用于人体细胞的?
A:通过扫描电子显微镜,深入到肺部的内皮,研究者发现实际的病毒颗粒在额外的细胞空间;如果你在显微镜下观察死于新冠人的肺部,你会看到,这些黑点都是渗透到肺中的淋巴细胞,使肺变得更硬,并由于这些淋巴细胞的免疫浸润而导致组织炎症的破坏已经到了内皮空间。当他们观察感染SARS-CoV-2或COVID-19的人时,他们发现所有患者都携带了识别SARS-CoV-2尖峰蛋白的辅助性T细胞,这是病毒用来感染细胞的蛋白质。
它们还含有辅助T细胞,对其他SARS-CoV-2蛋白起反应。 我们所说的是SARS-CoV-2中有与其他冠状病毒相匹配的蛋白质。在COVID-19患者中发现“T细胞对长期免疫有良好的预兆”。 就像我们之前讨论过的的T细胞、记忆性T细胞、辅助性T细胞、细胞毒性T细胞——这些细胞与长寿免疫有关。 因此,重要的是要看看他们对SARS-CoV-2的反应是否涉及T细胞。 所有的疫苗都在试图确保B细胞产生抗体,这实际上是我们正在测试的抗体,但适应性免疫系统的另一个重要部分是T细胞。 所以让我们看看他们在这里说什么。 他们谈到了免疫战士T细胞,帮助对抗一些病毒,但他们发现的两项研究表明,SARS-CoV-2感染者携带的T细胞实际上是针对病毒的-并可能帮助他们恢复。
这个是相关内容医学视频讲座的链接,需要可以持续关注:这个是相关内容医学视频讲座的链接,需要可以持续关注:



Add comment