Update 95: Widespread Thrombosis on Autopsy; Prognostic Factors
Medcram.com welcome to another medcram covid-19 update want to let everyone know that I took a little bit of a break last week. I’m feeling just fine and I do appreciate everybody’s concerns. We’ve got a couple of papers. I wanted to analyze and we’ll jump right into those right away. Let’s quickly take a look here around the world daily cases in the United States continue to climb daily Des are now starting to climb in the United States.
Interesting in California, the daily cases have kind of plateaued daily deaths in California are on the increase.
In Florida, there’s been an increase in Daily new cases.
And that is accompanied by an increase in Daily deaths in Texas. There is an increase in Daily new cases and this is accompanied by an increase more than doubling of the daily deaths in Texas.
We’ve been watching Australia it’s winter down there and we can see that there is an increase in the daily new cases. Currently the deaths in Australia are negligible India here definitely uptick in Daily new cases that has been very consistent here over the last number of weeks and we’ve seen a consistent increase in the daily deaths as well.
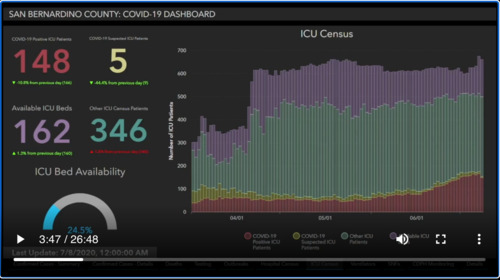
I want to show you what we’re working with here locally where I work in San Bernardino County on a regular basis and you can see here we’ve got what’s going on in the Intensive Care units across the County of San Bernardino, which is in Southern California. And here we have April 1st May 1st, June 1st, and we’re ending up here with July. I think this last one here is July 8. That’s the one that we have data for and we can see here what’s going on in the Intensive Care Unit in terms of ICU census these
Purple lines represent the available Mt. ICU beds. You can see here that in the beginning of April. We increased our capacity countywide to take care of patients. You can see if we hadn’t done that. We would be in Dire Straits at this point the green which is by far. The largest proportion of ICU beds. Here are actually other non covid ICU patients and you can see that that sort of started to come up once the opening up of
In California came to fruition here at the red going across our actual covid patience and this yellow as we go across here are suspected covid patients. You can see what the in provement in testing that has occurred over time that proportion has dropped dramatically and we’re now able to see exactly which patients are suspected and which ones are not suspected but clearly you can see that the number of ICU patients has gone up since June.
Pretty significantly almost a doubling of the number of ICU covid patients and overall. This represents may be either a quarter to a third of the number of ICU patients total in the entire County. You might think that that’s not a big deal, but if because of covid and when covid spreads it spreads exponentially it’s not going to take very much time to double if you were to double the amount of patients in the Intensive Care Unit that would completely use up all of the Surplus ICU beds that are available.
That point all patients requiring ICU care not just covid patients. But even those patients here that are non covid patients would feel the squeeze in terms of ICU beds. And so if you had a heart attack or if you had a pulmonary embolism or if you had the post-operative sepsis, it would be very difficult to find intensive care services, even though you don’t have covid-19. So this is just one snapshot here of one County in one state of the United States, which is just one country.
This is being multiplied in many many different counties and states and countries around the world. One of the questions might be how did you go from here available ICU beds to up to here as the amount patients requiring ICU care jumped. We have to be very creative. So what are we doing in these situations? Well in these situations the county and the state specifically is allowing us to license ICU beds where we would not be normally licensing ICU beds in the hospital. And where would that be?
That would be in regular patient rooms for instance in rooms. That would not be ICU. We could also be doing this in what we call the pacu. These are where patients come out of surgery and are staying and they’re being monitored their we could turn those into ICU beds. Of course, it’s not ideal. Sometimes the monitoring is not as good as it would normally be but as we’ve said many many times here the good is not the enemy of the perfect.
I want to talk about this paper because we’ve talked extensively in the past about the pathophysiology of covid-19. Don’t want to take time to go through a lot of that at this point. But remember that the concern is that the virus attacks the ace2 receptor which causes oxidative stress which causes inflammation of the endothelium releasing procoagulant substances, and that leads to thrombosis. Well here is a study out of New York.
Varsity looking at seven autopsies of patients and characterizing what they found and what they found was not just clots in the lungs which is what we’ve been looking at before there. Is that paper remember in the New England Journal of Medicine that looked at micro thrombi in the lungs, but here we’re seeing megakaryocytes. So what does it megakaryocytes megakaryocytes are the precursors to platelets? These are large cells Mega meaning large that have multiple aggregations if
If you will of small platelets coming into one cell. This is how they are made and we’re seeing a lot of these as well as platelet and fibrin thrombi not just in the lungs, but on these autopsy Series in just about every other organ in the body and the question is is are they forming there or the embolizing or traveling to those areas after they’ve been made so let’s take a look at this article that was published in the Lancet specifically eclinical.
Listen, they say here that there’s an increasing recognition of a pro thrombotic state in covid-19 and post-mortem examination can provide important mechanistic insights. So what they did was they looked at covid-19 autopsy Series in seven patients and looked at the lungs heart kidneys liver and bone at a New York academic Medical Center. This was NYU. So they found seven patients and about half of them were male half female and they looked at him regardless of anticoagulation.
So some of these patients were on anticoagulants. Some of them were not some of them are on full dose anticoagulation as we would call it. Some of them were just on prophylactic or preventative dose type of anticoagulants and notice here that all of the autopsies demonstrated platelet-rich thrombi in the pulmonary. That’s the lungs hepatic. That’s the liver renal kidney and cardiac microvasculature and that megakaryocytes. Those are those cells that give rise to platelets. We’re seeing in higher than
Usual numbers in the lungs and the heart and in two cases had thrombi in the large pulmonary arteries or casts conform to the anatomic location. So in other words it completely filled up the tubes. Let’s go on and take a look here and their interpretation they say in this series of seven covid-19 autopsies. Thrombosis was a prominent feature in multiple organs and in some cases despite full anticoagulation and regardless of timing of the disease course suggesting that throw
Bose is plays a role very early in the disease process the finding of megakaryocytes and platelet-rich thrombi in the lungs heart and kidney suggest a role in thrombosis here. We can see different tissues of the human body here in panel a this is the medium size blood vessel in the lung and we can see a coagulation here by the yellow star in be for those who are not inclined to know this. This is the kidney as you can see here.
Some tubules and you can see that there are at the arrows here thrombi in the kidneys here in see this is what the heart looks like under Section. You can see the the sarcomeres and the muscles fibers going across and here at the blue arrow. We see another thrombus occurring and then over here is lung tissue and microvasculature. We can see multiple blood clots here in the microvasculature. Here’s some more images from the study. This is the
the hilum of the lung and this is the pulmonary artery. These are clots that are sitting in the pulmonary artery preventing the blood from flowing and that they’re quite easily removed. You can see here in section be here in another case some more blood clots here. And when we section this we can see that the alveoli here instead of being nice and thin allowing the oxygen to diffuse into the blood has something called diffuse alveolar damage or d a d and you can see here in this
Artery that it is completely filled up with micro thrombi right where the yellow star is. Here’s another look at some more cases. This is the heart here. We have a micro thrombi with fibrin and thrombin but in the middle of it, you can see a megakaryocytes this make a carrier site will eventually break up into multiple platelets, but very interesting to see a megakaryocytes in this position. Not normally seen Carol. We see in slide B. This is a cross-section of
Of lung again, very thickened alveolar walls not very conducive to transmitting oxygen and what we see in all of these capillaries is we’re all these black arrows are our megakaryocytes. These are things that come from the bone marrow that normally break up into forming little platelets here. Those of you who can recognize this. This is kidney. You see a lot of microtubules and things of that nature and a glomerulus. That’s the big thing right here.
What do we see in the middle of the glomerulus again? We see something that lodged in there called a megakaryocytes. Why is that there when we look at the bone marrow where the megakaryocytes are actually coming from you can see some fat. Those are those round empty spaces because they get dissolved when this thing gets processed. But what we’re standing for hear something called CD 61, which we see on megakaryocytes and you can see that it is very rich in these megakaryocytes. Here’s some more slides related.
Mostly to the heart the red is muscular tissue. This is from the heart but you can see generally speaking a kind of a blackish Haze here. That is because what you’re seeing there are the nuclei of white blood cells. Yes, there’s diffuse inflammation here or myocarditis. And that’s what we’re seeing here is a bunch of white cells that are infiltrating here. And that’s what’s causing inflammation. We see it again over here in the epicardium. That’s the outermost layer of the heart and you can see that
Blue haze all through here. Those are all white blood cells that have gotten in there. If we look at the micro vasculature of the heart these little red dots that are pointed out by yellow arrows again megakaryocytes. That’s because we’re staining for CD 61 there. Why are they there? That’s a good question here. We see a cross section of the ventricle of the heart and you can see is some pallor of The ventricle and that may be related to less blood supply from ischemia here. We see a cutting open of
The heart and we can see in the trabeculae carne which is basically another way of saying just in the area here that tethers the mitral valve down and it’s sort of at the apex of the heart. We can see some clots also some clots over here in this area. This is fibrin clotting his another slide looking at this. Here’s the EKG on admission of another patient and the EKG a little bit later. Let’s zoom in on those so we can see that a little bit better.
for those who don’t know this 2 3 and avf look at the inferior aspect of the heart for definitely seeing ST segment elevation above the Baseline and also in V5 V6 and even V4 we can see that the ST segment is well above the Baseline indicating that this patient is having an ST segment elevation Mi and furthermore what the authors are trying to show here is a bit of hemorrhage at the junction of the left and right ventricle that may have caused some of
Findings on the EKG here again. We see another megakaryocytes in the strands of the fibers here of the heart. We see here at EF and G thrombus thrombus and thrombus all of those could be the reason for the myocardial infarction or the heart attack.
So what is there discussion of this? Let’s look at the first paragraph. They say in these series of seven autopsies in patients who succumb to covid-19. There are several unifying themes observed in the histopathology thrombosis is a prominent feature in multiple organs and in some cases. This is despite full anticoagulation. And this really goes along with what I’ve seen clinically in the patients that I have been taken care of despite them being on full dose anticoagulation. We’re still seeing complications of them having
Lots and what kind of clots are they they go on to say platelet-rich thrombi were present in the pulmonic hepatic renal and cardiac microvasculature based on the anatomic cast pattern in the thrombi many of the thrombi appear to have formed in situ. That means right there in place. In other words. They didn’t form elsewhere and travel to that place. They formed right there in that place. They also say that it was ante-mortem. What does that mean? That means there before they died. How do we know that?
It’s because of these lines of zon lines of zon as we showed you before are these layers that form and show that the blood clots formed while blood was pumping while there was fast flowing blood and so obviously that’s going to happen before somebody dies not clotting after they die. However, they say here that embolism with further local propagation cannot be excluded based on the lack of deep vein dissection other words. They didn’t look at the Deep veins in the autopsy to
If there was blood clots in those lower areas, they go on to say that the extensive nature of platelet fibrin thrombi in the alveolar capillaries in our patients may explain the observation that oxygenation is disrupted in an exaggerated fashion early in the disease course of patients with covid-19 as this suggest evidence of ventilation/perfusion mismatch unrelated to hyaline membrane formation. What are they saying here? They’re saying that yeah the alveolar membrane thickens, but
That’s probably not the problem and the cause of why we’re getting oxygenation issues. They’re thinking that maybe the oxygenation issues are related to these platelet fibrin thrombi and that’s what we’ve been saying now for a long time.
They also go on to mention the interesting finding of megakaryocytes both in the throng buyers themselves, but also significantly elevated in the bone marrow.
Now here’s the interesting paragraph what they say here is that the thrombi that they saw were located in the veins and in the pulmonary arteries and the pulmonary arterial roads and in the microvessels, but not in systemic arteries what they mean to say here is not in the arteries coming from the left ventricle that Supply the organs watch what else they say despite elevated fibrin degradation products in only one case of a patient with cirrhosis. Did we
Our glomerular thrombotic microangiopathic. Why is this interesting this would seem to go against the possibility that this is D IC or disseminated intravascular coagulation hemolytic uremic syndrome or thrombotic thrombocytopenic purpura as the predominant pathophysiological pathway. Now that’s important for those that understand these things because these things are one of the leading suspicious characters or
Processes that could be causing this schistocytes are simply just broken up red blood cells as the red blood cells are careening through these vessels when there are little strands of fibrin, they get sheared on these and they get destroyed instead of seeing nice round red blood cells. You see fragments of red blood cells and those are known as schistocytes. Well, those are typically seen when you have endothelial damage. They didn’t see any of those they found no endothelial abnormalities now that would seem
the fly in the face of what we’ve been saying which is that endothelial damage because of the tsar’s cup of to virus binding the H2 receptor causes oxidative stress and the filial damage and then thrombosis but notice what they say here, but we cannot rule out increased exposure of tissue factor a Roshan of the endothelial glycocalyx or other mechanisms of endothelial dysfunction that could be procoagulant without showing histopathologic evidence of Cell Activation or
Ian a recent autopsy series of covid-19 cases Incorporated and the filial electron microscopy demonstrated ultrastructural and ethereal damage and increased angiogenesis as compared to cases of fatal influenza with differential regulation of genes related to angiogenesis and inflammation. They go on to talk about troponin and elevated troponin Xin patients with covid-19. And why might that be and one of the theories is that there might be viral myocarditis, but they said that this is
Actually a pretty rare occurrence and in fact a previous covid-19 patient that did have viral. Myocarditis was actually infected with influenza at the time. So it’s hard to say they conclude that tissue proven viral. Myocarditis appears to be a relatively rare occurrence in covid-19. They feel that based on their data from both sides of the micro vasculature and the cardiac veins appears to be a common cause of elevated troponin.
And covid-19. If you had thrombosis of the cardiac veins that would mean that the blood would not be able to drain back into the venous system. And that would cause a pressure load and therefore inability to get oxygen to the cardiac muscle which would cause an elevation of troponin. In fact the occurrence of thrombosis and covid-19. Apparently is so common in these series of covid-19 autopsies that they were able to demonstrate for the first
I’m in one of their autopsies that there was a thrombosis of a cardiac vein not a cardiac artery but a cardiac vein and that this could cause acute myocardial infarction in the absence of obstructive coronary atherosclerosis. In other words. It’s very possible that you may have a patient with covid-19. They have an MI and you send them to the cath lab afterwards and you do the cath lab you do an angiography and their arteries are beautiful. There’s no thrombosis anywhere. Well it could
Very well be that the thrombosis is not in the coronary arteries, but in fact in the coronary vein.
In one of their cases here in case number three, they had a thrombosis of an intra myocardial vein corresponding exactly to the infarction Zone.
In terms of the kidney there was virions that were actually observed in the proximal tubular cells. And of course a number of acute tubular necrosis at various different levels were seen in a number of patients in previous studies. So all in all are very interesting study of course will put the link to it in the description below. But I want to take this with these findings in mind and show you a second study, which I think will be very useful.
Practically for those of us taking care of patients in the hospital and that’s this paper titled prognostic factors associated with mortality risk and disease progression in 639 critically ill patients with covid-19 in Europe initial report of the international risk 99 ICU prospective observational cohort and this paper was published out of Zurich, Switzerland.
So what they did here was they took 639 critically ill patients looked at a number variables and they try to determine which things were going to determine likelihood of survival versus non survival and what they came up with was that the patient’s creating D-dimer lactate potassium PF ratio a a gradient and ischemic heart disease, all of those were independently associated with ICU mortality.
So let’s take a look at those findings. And as you can see here, they grafted out looking at day 0 all the way to day 7 and most of these cases and here you can see on the y-axis things like leukocytes the neutrophil / the leucocyte ratio thrombocytes things like the D-dimer level interleukin 6 plotted over time. So the nonsurvivors are in red and the survivors are in blue.
And I did a bunch of statistical analysis to see which ones could be seen as independently determining whether or not the patient was going to survive or not survive. So let’s take a look at those.
here they are so again we have overall survival in days and these are kaplan-meier survival curves by the way so you can see here that we start off at one point zero zero and the red and the blue here are different they determine what the variable is so for instance this one is the correct meaning and its measured in micro molar per liter but if we want to convert that into what we would see normally in the United States then we would say that a creatinine of less than
A hundred or greater than or equal to a hundred would be around 1.14. So that would be the cutoff if you want it to make this practical you can just say one point one. So if patients had a cracking of greater than one point one, then they would be in this red line versus those having a cracking of less than 1.1 which would be here in this blue line having a slightly higher but statistically significant survival rates.
And you can see here that the hazard ratio is two point zero nine. Well, let’s take a look at some of the other ones here here. We have D-dimer if it was less than fifteen sixty, then you would be in the blue higher chance of survival greater than fifteen. Sixty lower chance of survival. These are all basically on admission here we have do you have ischemic heart disease or do you not have ischemic heart disease clearly here? If you said no better chance of survival. If you said yes worse chance of survival.
Take a look over here at the lactate. If you are less than one point five five better chance of survival on admission here versus greater than 1.5 V less chance of survival. What about the potassium if the potassium was less than 4.16 better chance of survival versus greater than 4.16 and finally the PF ratio. And for those who don’t remember what the PF ratio is it simply the pao2 on the blood gas.
And by the amount of oxygen that the patient is on so for instance if the pao2 is 80 and the patient is on 50% fio2, then it would be 80 divided by point 5 which would be 160 and we would just look at this here. And of course if it was less than two hundred and six less chance of survival greater than 206 on admission higher chance of survival. So if you look at these there’s a case to be made in each one of these situations.
Of micro thrombi if there is micro thrombi in the kidney that’s going to make the creatinine worse. Obviously at the micro thrombi is potentially worse. We’re going to have a higher D-dimer level if we’ve got micro thrombi. Somebody with ischemic heart disease is not going to tolerate that as well here again with a lactate if we have Micro thrombi potentially, we’re not going to have perfusion and oxygenation if we have no oxygenation or less oxygenation that
That means we’re going to have an aerobic metabolism and the product of anaerobic metabolism is lactate again potassium may be just simply a surrogate here of the ability of the kidney to process potassium. If it’s not processing it, well, you’re going to have higher potassium rates. And then finally the PF ratio is pretty self-explanatory. If you have Micro thrombi in the lungs, your PF ratios are going to be very low so you can see here that in each one of these situations. It could independently lead to a worse survival.
Based on thrombosis alone, but I believe the one that is closest and a direct measurement of micro thrombi is in fact the D-dimer and if you’ll notice the hazard ratio out of all of these is the highest in the D-dimer. So I think that is probably the most important you can also see that it has the biggest difference between survival and nonsurvival in any of these independently linked Hazard ratios.
So I thought this was an excellent article. I believe that it is casting light on to what is really going on here in covid-19 in terms of the pathophysiology. I believe based on what we’re seeing here that thrombosis particularly with platelets and fibrin and megakaryocytes is what is leading us to the hypoxic mechanisms with covid-19. I believe it is happening very early in the course probably.
Before the patients even get to the hospital.
I also believe it’s probably the reason why we’re seeing a very wide range of symptomatology in patients because it really just depends on where the thrombosis occurs if the thrombosis is occurring in the brain, you’re going to get neurological symptoms if the thrombosis is occurring in the lung, you’re going to get shortness of breath symptoms if the thrombosis is occurring in the kidney, you’re going to get renal issues and I think more deliberate research and really needs to go into how do we prevent these?
Catastrophic micro thrombi from occurring. Thanks for joining us.



Add comment