Testing; Causes of Hypoxemia in COVID-19 (VQ vs Shunt vs Diffusion) [Lecture 58]
本文由‘中国推动’学者、济南大学生命科学学院生物技术专业任紫豪同学整理。
Welcome to another Medcram COVID-19 update. I’ve got a lot of news to cover today.
The United States tops the charts with the number of new deaths at 1867. United States daily new cases is hovering right around 30,000. Also United States daily deaths around 2,000 doesn’t seem to be dropping much at this point. Likewise, daily Canadian new cases around 1500 and it looks like Canadian Daily deaths are not peeking as yet or stabilizing. It continues to increase. United Kingdom seems to be hovering around 5,000 cases a day. Daily deaths of the United Kingdom are pretty constant here around 750.
I wanted to talk a little bit about testing. And again, I want to review there’s different types of testing that’s going on up to this point. We’ve looked at Reverse Transcriptase PCR and here we’re looking at the RNA of the virus itself. This is usually done by a nasal swab and if it’s positive it tells you that you are infected and if it goes from positive to negative, generally speaking, what you’re trying to find out is whether or not you’re still infective.
Now this is very different than antibody testing. So with antibodies you have a antigen presenting cell that has eaten an infected cell and is presenting a portion of the virus on its surface that connects up with a T cell. And once that connection is made, there are cytokines which induce that T Cell to do a number of things. Number one, the first possibilities are memory cells. Number two are cytotoxic T cells, and number three are B cells that turned to plasma cells and they create things called antibodies and these antibodies go out and attack the virus. So this is different. We’re not looking for RNA of the virus. We are looking for proteins or specifically antibodies. This is not done with the nasal swab but it’s done with the Finger Prick and what this tells you is whether or not you have IgG or IgM.
IgM and IgG usually come up maybe around 6 to 7 days, generally speaking, after the beginning of an infection and they both sort of come up but the IgM will go away and the IgG will be the only one that remains usually and so IgM is usually seen in acute and IgG is chronic. One of the things you should understand is just because you have the presence of antibodies. It doesn’t mean that you would be negative on the infective state. So it’s possible to be both positive on the Reverse Transcriptase PCR and also be positive on the antibody testing and that would simply mean that your body has made antibodies against the virus, but you still have the virus and you’re still infective.
So understanding that it’s very important when we read articles like this in the Boston Globe titled nearly 1/3 of 200 blood samples taken in Chelsea show exposure to coronavirus. Mass. General researcher says that the results point to a raging epidemic, but may also indicate the city is further along the disease curve than some other municipalities.So what they did was they went down to Bellingham square and they tested about 200 Chelsea residents there and they found that 64 of those residents were positive. They made sure to exclude those people that have been tested positive using an RT-PCR tests prior.
But it’s important to understand that this was not a random sample. No, in fact Chelsea has the state’s highest rate of confirmed COVID-19 cases at least 39 residents have died from the virus and 712 tested positive. As of Tuesday, at a rate of about 1,900 cases per 100,000 residents, or almost 2%. So why would there be 30% in this sample? As we just said, this is not a random sample, so it may not be representative, but boy a third of cases sounds pretty high. Now as we’ve mentioned before just because you have an antibody that’s positive, it doesn’t mean that you’ve cleared the virus.
Still, Mr. Ambrosino who is the city manager added here, it’s kind of sobering that 30% of a random group of 200 people that are showing no symptoms are in fact infected. It’s all the more reason for everyone to be practicing physical distancing. They also go on to talk about that many work in the hospitality industry and health-related fields where exposure to the virus is greater. And a lot of them must go to work during the pandemic. Just to know that the test that they used here was by BioMedomics, which is not yet FDA approved, but that Mass. General determined that was reliable.
Here was an article that was published on Med Archive that has not yet been peer-reviewed that looked at a slightly different situation on the West Coast.
In this situation, they reported the prevalence of antibodies to SARS-CoV-2 in a sample of 3,330 people adjusting for zip code, sex, race and ethnicity. So what they found was after they did that adjustment, the unadjusted prevalence of antibodies to SARS-CoV-2 in Santa Clara County was 1.5% or somewhere 1.11-1.97% and that the population-weighted prevalence was 2.81% and what they came up with was that this was 50-85-fold more than the number of confirmed cases. And so their conclusion was that the population prevalence of SARS-CoV-2 antibodies in Santa Clara County implies that the infection is much more widespread than indicated by the number of confirmed cases.
Population prevalence estimates can now be used to calibrate epidemic and mortality projections, but there’s an interesting pair review that was published on medium.com of this article by researchers at Stanford University.
He talks about how he really hopes that the article is true, because of course if it is that widespread then that means the fatality rate is actually a lot less that there may be more people than we think that are infected and this would be closer potentially to widespread immunity and it would allow people to go back to work. But he talks about why he is skeptical of this research article for several reasons, and he enumerates these reasons here.
The first one being that false positive rate of the test is actually too high. And basically he says that if the noise from the false positives are high enough, it could actually bury the true false positive rate and make it look as though the number of positives are higher than they really should be. And he goes through and shows us what the definition of a false positive is, but what it really boils it down to is apparently what a gentleman by the name of Jeffrey Spence says, he says here, in the supplement they say that 2 out of 371 plus 35 known negative samples tested positive. This means that the 95% confidence interval for the false positive rate is somewhere between 0.06% and 1.77% and in their samples from Santa Clara County, they had 1.5% test positive. So you see here that the 1.5% fits within the 95% confidence interval that is given for our false positive rate and so what you can see here is that they can’t say with 95% confidence that the positive rate that they got from their antibody tests isn’t all from false positives. And what Dr. Srinivasan who is the PHD author of this peer review is saying here is that Jeffrey likely means that 371 plus 30 no negatives here rather than the 371 plus 35 and our confidence interval somewhat differs from his but the conclusions are directionally the same.
And the author shows this through three progressively more sophisticated ways to estimate the false positive rate, which he lists here and he starts with these three and it’s maybe worth your time to look through but probably beyond the scope of what we’re talking about here.
Finally, he boils it down to this. The main conclusion is that we don’t have a high confidence that the false positive rate is low enough and that this is one possible failure mode. The second point he makes is was this really a randomized sample. And in fact what they did was they recruited participants via Facebook ads, which if you haven’t had any symptoms you probably wouldn’t care but if you had had symptoms and you wanted to find out whether or not you might have had a COVID case maybe a month ago, you might be pushed to have an antibody test and that’s exactly the point that he makes here.
He says here, but there’s a problem:what if their group of participants was enriched for positives relative to the general population? What if their participants had a much higher rate of COVID-19 than normal? And as he says to the author’s credit of the original article, they did a test to this. They say that their study had several limitations. First, our sampling strategy selected for members of Santa Clara County with access to Facebook and a car to attend drive-through testing sites. This resulted in an over-representation of white women between the ages of 19 and 64 and an under-representation of Hispanic and Asian populations relative to our community. But the big Whopper here is here at the end, other biases such as bias favoring individuals in good health capable of attending our testing sites or, here’s the key, bias favoring those with prior COVID-like illnesses seeking antibody confirmation are also possible. The overall effect of such biases is hard to ascertain.
And apparently the authors of the original article didn’t feel that that was a big enough deal, but doctor Srinivasan, the author of this peer review, felt like it was a big deal. He says here the first mechanism that could significantly enriched the number of COVID-19 cases in the study is if symptomatic or expose people use the study to get a test they could get no other way.
And of course testing is pretty minimal right now in California. He states here after all in the Bay Area in early April it was really hard to get a test for people with mild symptoms or exposure. So people who thought they were exposed or symptomatic may have signed up for the study to get access to a free COVID-19 test they could get no other way and apparently all they needed was 50 out of 3330 to exhibit this behavior.
And then of course people who are exposed may have helped other people who they have direct contact with to get into the study who they could have exposed as well and instead of getting a super spreader you would get what he calls a super recruiter event where one person recruits N other enriched people into the study and that could significantly boost the number of positives beyond what you’d see in a random sample of Santa Clara.
So at the end he summarizes his conclusions as we talked about. The first one was the false positive rate. The second was there might have been an enrichment of COVID-19 cases in his sampling. The third is that it seems as though in order to produce the visible excess mortality numbers that COVID-19 is already piling up in Europe and New York City, the study would imply that COVID-19 is spreading significantly faster than past pandemics like H1N1, many of which had multiple waves and took more than a year to run their course.
So why have we taken so much time looking at this? I think it’s instructive because, number one the original paper that was about to be published or is on Med Archive does seem to make some very interesting claims. Claims that show that the virus is far more prevalent than we think. Number two, the peer review by Dr. Srinivasan is instructive in exactly why peer review is important. Sometimes these findings that we see may not be what they seem but what we will see is that time will tell. And usually the answer is not on one extreme or the other but somewhere in the middle, so I’d be interested to see what comments you have below.
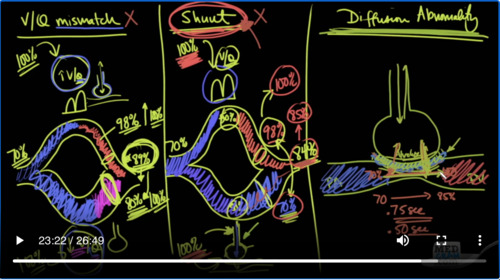
I wanted to switch gears a little bit now and talk to you about some of the mechanisms of hypoxemia in COVID-19 and in other diseases as well, and this has come up because there have been a number of discussions on the internet and on YouTube about Physicians talking about the mechanism of how people are getting hypoxemia with COVID-19. And in one video in particular the discussion is about how V/Q mismatch and Shunt Physiology may not describe exactly what’s going on in COVID-19, but that diffusion abnormality seems to be a better explanation. So if you don’t understand what those things are, you’re going to be completely lost when it comes to discussing this and being able to entertain that kind of language. So I wanted to quickly go over exactly what is V/Q mismatch, Shunt and Diffusion Abnormality. So with V/Q mismatch one way of looking at this is looking at the blood that is going to the lungs.
It’s in areas where there is high V/Q relationships and areas where there’s low V/Q relationships, and I’ll explain a little bit more about what that is. So you have to understand that with an alveolus. It’s very important that the amount of ventilation that’s going in and out of the alveolus is commensurate with the amount of blood that’s going by. If there is a difference between the amount of air flow and the amount of blood flow that’s going to cause problems.
Now there always has a slight maldistribution in the lung but it’s a very very small amount what can happen though is specially in thrombotic problems where there is blood clots is you can get issues with V/Q mismatch. Of course, you can get it with other diseases as well. But here’s an example.
So I’ve drawn this to show you that when deoxygenated blood is coming in to the lungs, if there are areas that have more blood going to it and there are areas with less blood. By definition, Q is flow. And so if there’s an area here of less blood go into this ventilation units, then your V/Q are going to be high. Here on the other hand, if there’s more blood coming in and that’s going to increase the denominator, then your V/Q is going to be low. So you have here is a high V/Q ratio and a low V/Q ratio. And so therefore you have mismatching V/Q and so why would that cause hypoxemia? It’s simple it’s because in this blood coming back, that’s very well-ventilated given the amount of blood that’s coming back. This is going to be very well-oxygenated.
However, the same cannot be said here in this situation you’re going to get oxygenation but not as much because there’s less of ventilation per perfusion. And so here 70% saturation is what the blood usually is when it comes into the pulmonary artery when it goes to the pulmonary vein and have oxygen or blood coming back. You might say for instance here have 98% saturated blood because it’s very well-ventilated but not very well-perfused. But here you have a very large amount of blood coming back which is not very well-ventilated. And so here it may only be let’s say 80% saturated.
Well, when this blood comes back together, it’s going to be normally just an average of these two, but it’s going to be the weighted average based on the blood. But because the amount of blood coming back here is so small and the amount of blood coming back here is so large that it’s not going to be the arithmetic mean of these two saturations. In other words. It’s not going to be 89%. No, no, no, actually it’s going to be less than 89%. The reason is that because this has more flow and because they are that has more flow will always have the lowest saturation. The average of these two is always going to be closer to 80% or the lower range. Now if we give 100% oxygen to these patients here and here, you can see very quickly that not only is this 98% going to go up to maybe100%. So that’s not a very big deal there but here because there is ventilation because we can get oxygenation into them. We’re going to get 100% here and here and so it is possible to improve the oxygen saturation by giving patients 100% oxygen.
This is encounter distinction to Shunt Physiology. Okay, let’s show you what’s going on here was Shunt Physiology. Same sort of situation, start out at 70%. And in this case, we’re just going to say that the Shunt Physiology is about 50%, so 50% is going here and 50% is going here. Now in this situation, we have the lungs up here and let’s just say that the V/Q ratios are just fine. And so 70% blood coming in from this side is going to go here and because it’s going to the lungs it’s going to get well-oxygenated and it’s going to come back saturated at 98%, whereas here this blood coming in to this limb is going through a shunt. What does that mean?
Well, there may be an alveolus that’s here that air could get into and could oxygenate the blood going by but because there’s fluid in here either proteinaceous fluid or it’s just collapsed. There is absolutely no way that that are or that ventilation can get to that area where it can get into the circulation and so as a result of that this blood simply goes right back at 70%. In other words, there’s no oxygenation that occurs. It comes in at 70 and it leaves at 70, whereas this could goes to the part of the lungs that are being ventilated appropriately will be resaturated at 98%. So, because this amount of blood is equal to this amount of blood because we said it was 50%, 50% shunts. This will be the arithmetic mean of these two.
So in that case since there’s a difference of 28 points, then this will increase by 14 points and the new saturation will be 84%. Let’s go ahead and put 100% oxygen on this patient, 100% here and 100% here. Because the ventilation is not getting to it anyway, it’s going to be not consequential at all that the patient is getting 100% oxygen. It’s getting oxygen here, but it’s not able to go anywhere. So therefore this will still remain at 70%. It’s completely blocked, whereas up here this is getting ventilated appropriately.
And so, you’re going to super saturate this blood and instead of 98% it’s going to go up to 100%. Great. Now let’s do this calculation, 100%, 70%, now instead of 84%, it’s going to be 85%. And you can see that that’s just a very modest improvement in oxygenation, very very small amount of improvement in oxygenation. So one of the key things that you should know about Shunt Physiology is that when you give them 100% oxygen, it should not really improve oxygenation.
What does improve oxygenation in Shunt physiology is not increasing the amount of oxygen that you’re giving patients, but in fact the amount of pressure that you’re giving it under. So if you’re giving high flow or if you’re giving keep on a ventilator, then that could potentially pop open very nicely those alveoli and allow ventilation to go in and down. And now you can actually oxygenate and ventilate these alveoli to the point where you can improve oxygenation. That tells you that it’s shunt phenomenon. So shunt phenomenon, Again, the characteristic is that increasing oxygenation will not change the resulting saturation but increasing positive pressure will do that.
However, what a lot of these Emergency Room Physicians who are on the front line seeing these patients are saying is that patients are coming in extremely hypoxic. They’re being put on oxygen and they are improving so they feel that shunt physiology doesn’t explain it as well. And they’re saying that maybe V/ Q physiology mismatched doesn’t explain it.
So what some people have proposed out there is Diffusion Abnormality. So what is the Diffusion Abnormality? It’s very simple, diffusion abnormality is when you have an alveolus and you have a pulmonary artery going to a pulmonary vein through a capillary. So this is the pulmonary artery. This is the pulmonary vein. And it remember again that the pulmonary artery has deoxygenated blood and that is going to oxygenated blood and here you have an alveolus and you’ve got ventilation you’ve got carbon dioxide and oxygen coming in.
But what you have here is this very thin layer which is made up of the cell bodies of the endothelium on this side and the epithelium on this side. So this is extremely thin and what can happen is if there is fluid sometimes fluid can accumulate in here and make it thicker and that can cause a diffusion out the melody. It’s, so it’s harder for oxygen and carbon dioxide to diffuse back and forth through this. Now as it turns out carbon dioxide is not as affected as oxygen because of its ability to diffuse much more quickly because of its molecular structure, whereas oxygen has a much harder time diffusing in these things.
And so if you slow down the ability of oxygen to diffuse, then it’s possible that the red blood cells as they are passing by will not equilibrate instead of having 70% here going to 100%. They may go from 70% to only 85%. Why? Because the red blood cell is going by too quickly. Normally speaking, just so you’re aware, in a resting situation the red blood cell spends about 0.75 seconds in the capillary membrane as it goes by. Of course, if you do exercise, the cardiac output is going to increase and the red blood cell is going to whip by even faster. And so it might spend only 0.5 seconds.
Now for equilibration to occur this can occur very very quickly if you have a very fine-tuned and thin barrier for oxygen to diffuse through and so you may only need 0.4-0.5 seconds for oxygen to diffuse. But if you have a diffusion abnormality, which causes a barrier now it might take instead of 0.4-0.5 seconds, it may take a full second in some situations or 0.75 seconds for that oxygen to diffuse and if you start to exercise you can see that the red blood cell is going to spend less time because it’s going faster and it will not be able to completely take on all the oxygen that it should. So, that is diffusion abnormality.
And I would say that if you want to have a fuller understanding of these three and another two types of hypoxemia or the mechanisms of hypoxemia. Please go to medcram.com for a whole course on the different mechanisms of hypoxemia and that is completely free.
There are two more points that I want to make regarding all of this V/Q mismatch, shunt and diffusion abnormality. The first point is you should not fall into the trap of thinking that one type of disease only causes one mechanism. Let me give you an example. For instance, if somebody had mild congestive heart failure, mild congestive heart failure would first cause an accumulation of fluid right here between the alveolus and the pulmonary capillary membrane. And so it’s possible to have a mild diffusion abnormality in very early congestive heart failure. But as the congestive heart failure gets worse and worse and worse, not only will the fluid be in the capillary membrane, but if you look over here on shut it could fill up the entire alveolus. So it was completely fill a fluid when it’s more severe and then instead of having a diffusion abnormality, you would have a shunt phenomenon. So that’s just a simple explanation.
In fact in a lot of different diseases you might see a bit of diffusion abnormality, you might see some shunting you might see some V/Q mismatch, so that brings me to the second point. And that is this it’s possible to actually quantitatively find out exactly what’s going on in terms of V/Q mismatch shunts and diffusion abnormality. And the technique is called multiple inert gas elimination technique. This is where they put inert gases intravenously and they’re able to see how they are cleared. The description of how this works goes well beyond the ability to describe it here and in this forum, but it’s well noted here that in addition to quantifying ventilation perfusion inequality and pulmonary shunting, MIGET or multiple inert gas elimination technique can identify and quantify diffusion limitation of oxygen exchange, when present, as well as explain the contributions of extrapulmonary influences such as inspired oxygen concentration, ventilation, cardiac output, hemoglobin concentration, body temperature and acid/base state on arterial oxygenation.
So what I would propose to the scientific Community is to see if it’s possible to get one of these patients, one of these patients that many of the physicians have been saying are not acting like your typical ARDS patients to see if they could quickly identify who they are and do multiple inert gas elimination technique. But there is another point that I would make, this COVID-19 disease is much like an elephant in a dark room and we are trying to feel the animal to see what kind of animal it is and depending on where we’re standing in the room. It might feel like a tree trunk. It might feel like a rope. But at the end of the day, it’s all one animal. It may be presenting as different things to different people. Thanks for joining us.



Add comment